
Late on Wednesday night – here is the latest:
Arizona 5, California 12, Idaho 2, Illinois 2, Kentucky 1, Massachusetts 2, Maine 1, Michigan 1, Minnesota 3, North Carolina 2, New Jersey 1, Ohio 1, Oregon 4, Pennsylvania 1, Rhode Island 1, Texas 8, Virginia 1, Washington 2 and Wisconsin 1.
The FDA and CDC, in collaboration with the California Department of Public Health (CDPH), Infant Botulism Treatment and Prevention Program (IBTPP), and other state and local partners, continue to investigate a multistate outbreak of infant botulism. Epidemiologic and laboratory data show that ByHeart Whole Nutrition infant formula is contaminated with Clostridium botulinum, which is causing infant illness in multiple regions of the country.
ByHeart’s and FDA’s investigations into the root cause of the outbreak are ongoing, and at this time, FDA cannot rule out the possibility that contamination might have affected all ByHeart formula products. In response, CDC broadened the case definition to include any infant with botulism who was exposed to ByHeart formula at any time since the product’s release in March 2022. As of December 10, 2025, a total of 51 infants with suspected or confirmed infant botulism and confirmed exposure to ByHeart Whole Nutrition infant formula (various lots) have been reported from 19 states.
Previously, case counts included illnesses from August 1, 2025, onward. With the expanded definition, CDC and state partners identified 10 additional cases that occurred from December 2023 through July 2025. At this time, no cases have been identified between March 2022 and December 2023. All 10 are confirmed infant botulism cases with documented exposure to ByHeart formula.
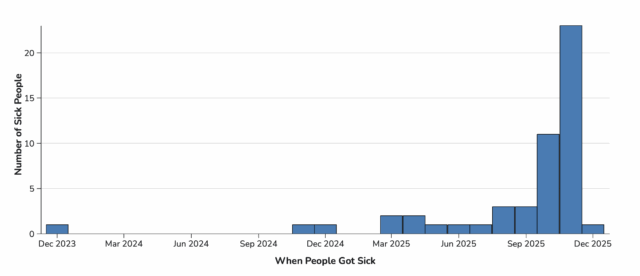
Illnesses started on dates ranging from December 24, 2023 to December 1, 2025. All 51 infants were hospitalized. No deaths have been reported to date. The infants range in age from 16 to 264 days and 22 (43%) are female.
FDA has not received reports of recalled formula being found on store shelves since November 26, 2025. All ByHeart infant formula products have been recalled, and these products should not be available for sale in stores or online. This includes all formula cans and single-serve “anywhere pack” sticks.
The ByHeart infant formula recall impacts markets outside the United States. Customer information provided by Amazon shows that a limited quantity of recalled ByHeart infant formula was distributed to Argentina, Brazil, Brunei, Canada, Chile, China, Colombia, Ecuador, Egypt, Hong Kong, Israel, Jamaica, Japan, Republic of Korea, Peru, Philippines, Romania, Singapore, South Africa, Thailand, and the British Virgin Islands.
Consumers worldwide should not use any ByHeart brand infant formula as all ByHeart products are included in this recall.